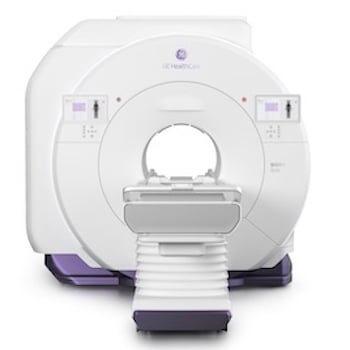
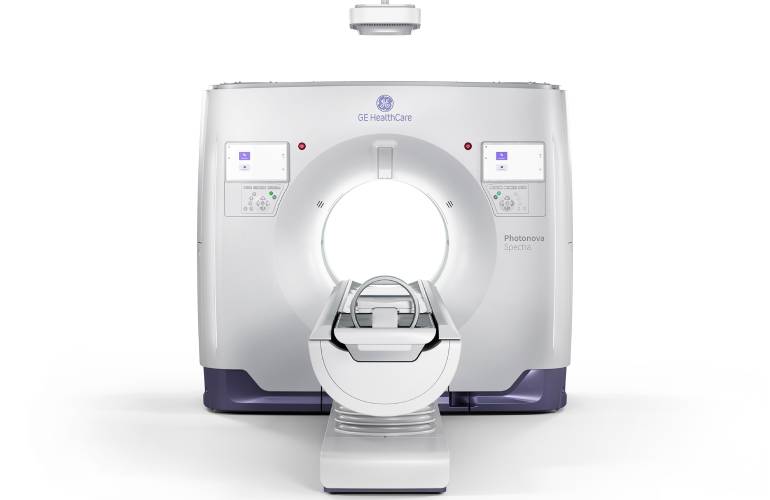
GE HealthCare submits AI-powered photon CT system to FDA
GE HealthCare
(Nasdaq: GEHC)
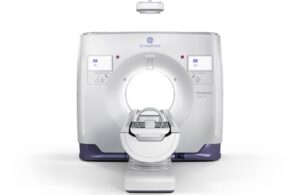
announced today that it submitted its Photonova Spectra system with advanced AI algorithms for FDA 510(k) clearance.
The company built its new photon-counting computed tomography (PCCT) system on its proprietary Deep Silicon detector technology. It designed Photonova Spectra to deliver “remarkable” spectral and spatial resolution for ultra-high-definition (UHD) imaging with wide coverage.
Unlike conventional CT converting X-rays into light before measuring, photon-counting CT directly counts individual X-ray photons. It measures their energy, enabling the potential for higher spectral and spacial resolution and improved tissue characterization. Deep Silicon, a novel detector material, enhances spectrial imaging, supporting advanced lesion characterization and treatment monitoring.
Chicago-based GE HealthCare aims to enable fast acquisition speeds, precise visualization of anatomical structures and enhanced material separation.
The company designed its system to address the challenges of fast-paced healthcare environments with rising patient volumes and diagnostic complexities. It says the on-demand spectral imaging can help detect, characterize and monitor disease with confidence. Staff can also utilize one protocol setup for many exams to reduce xomplexity and support efficiency.
“Today marks a transformative leap for GE HealthCare and a bold new chapter in CT innovation. Photonova Spectra is more than a new product – it’s a demonstration of what’s possible when vision meets purposeful design,” said Peter Arduini, president & CEO of GE HealthCare. “Built to give healthcare teams the clarity and confidence they need, this system aims to redefine decision-making and care delivery – meeting today’s challenges and tomorrow’s possibilities. This is innovation with impact designed to reshape workflows, sharpen image quality and empower confident, timely decisions. I am immensely proud of our teams and collaborators who are transforming the future of CT and bringing precision care to life.”
More about the new GE HealthCare imaging technology
GE HealthCare says that its silicon-enabled technology gives clinicians a way to obtain images with high contrast, low-contrast detectability and improved material characterization. It potentially enables greater diagnostic confidence.
The Photonova Spectra platform aims to enhance image quality by reducing signal overload and improving energy separation. It enables the photon-counting CT to clearly distinguish between materials like iodine, calcium and fat.
GE HealthCare says its system can enhance treatments in spaces like neurology, oncology, musculoskeletal imaging, thoracic imaging and cardiology. It can deliver visualization of tiny structures in the brain for neurology and characterize lesions in oncology. The system can visualize small fractures and bone marrow edema as well. Additionally, it offers ultra-high-definition chest scans and full chest imaging to enable in-stent lumen assessment, plaque characterization and myocardial assessment.
The company said its system can harness up to 50 times more data than conventional CT through Nvidia computing technology as well.
“Engineered with purpose to tackle some of healthcare’s most complex challenges – Photonova Spectra represents the next generation of CT innovation. From our earliest breakthroughs to today’s newest wave of innovation, GE HealthCare has innovated with intention, delivering technologies that empower clinicians and transform care,” said Roland Rott, president and CEO, Imaging, GE HealthCare. “As pioneers in AI and medical devices, we have harnessed advanced algorithms to materially enable this product – combining photon counting CT with our proprietary Deep Silicon detector. We are not just aiming to advance image quality, but to redefine diagnostic confidence and shape the future of precision medicine.”
link